Why Dry Pet Food Requires Preservatives
Commercial kibble is designed to remain stable for extended periods at room temperature.
This requirement creates a fundamental challenge: fat oxidation.
Rendered animal fats and lipid-containing ingredients oxidize when exposed to oxygen, heat, and light.¹ Oxidation leads to rancidity, nutrient degradation, and loss of palatability. Without preservation, commercial kibble (dry pet food) would deteriorate rapidly during storage and distribution.
Preservatives exist to slow oxidative reactions, not to improve nutritional quality.
Table of Contents
Oxidation in Rendered Fats
Most fat in kibble originates from rendered animal sources and is applied after extrusion as a surface coating.²
This fat is particularly susceptible to oxidation because:
Rendering exposes lipids to high heat
Surface application increases oxygen exposure
Long storage periods extend oxidative risk
The rendered ingredients in pet food oxidized fats degrade fat-soluble vitamins and generate secondary compounds associated with reduced food quality.³
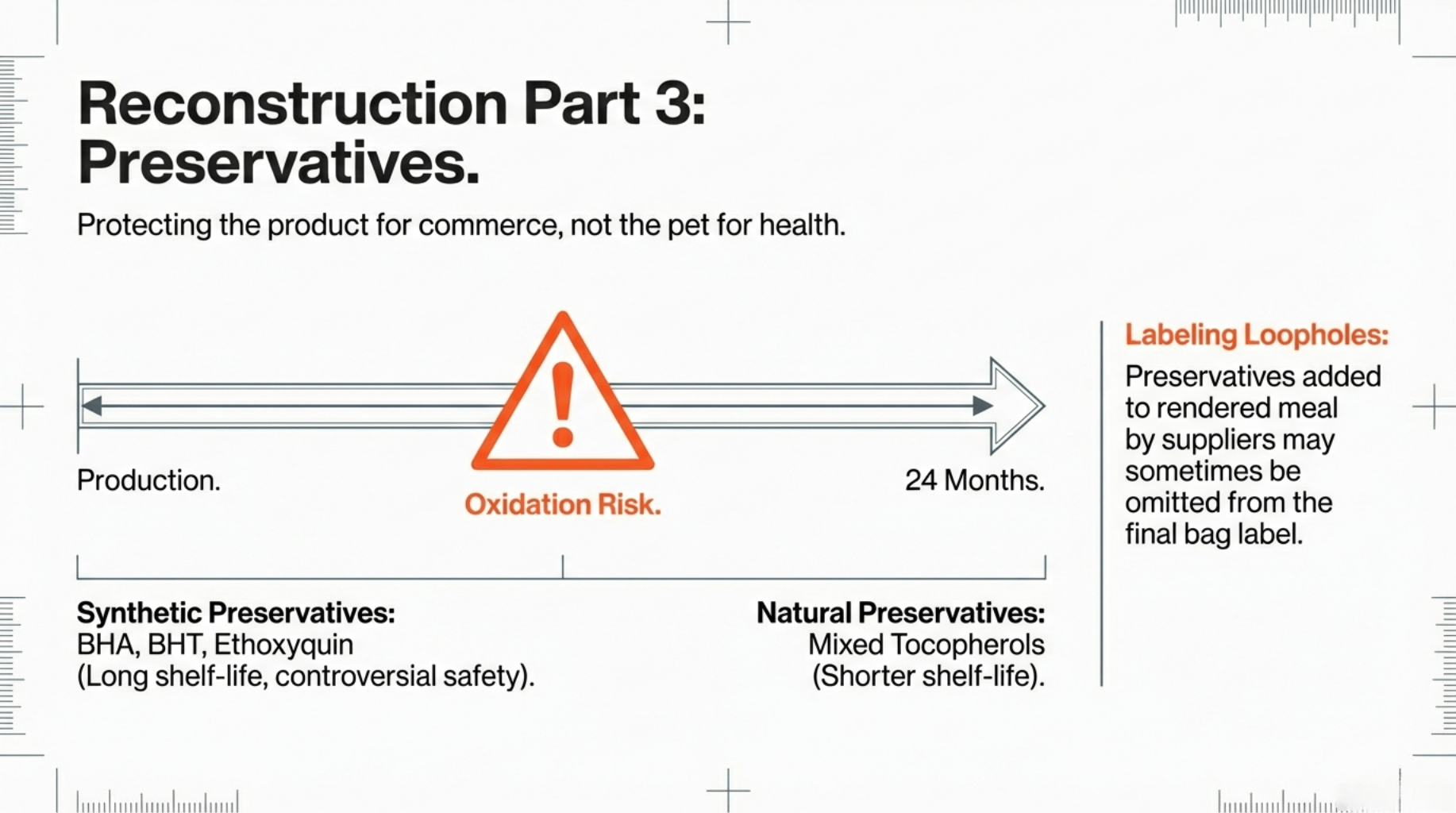
Types of Preservatives Used in Kibble
Preservatives in pet food fall into two broad categories: synthetic and naturally derived antioxidants.
Synthetic Preservatives
Naturally Derived Preservatives
Common synthetic antioxidants include:
Ethoxyquin
Butylated hydroxyanisole (BHA)
Butylated hydroxytoluene (BHT)
These compounds are highly effective at preventing oxidation at low inclusion rates.⁴ Their use is regulated with defined maximum levels.
So-called “natural” preservatives include:
Mixed tocopherols (vitamin E)
Ascorbic acid (vitamin C)
Rosemary extract
These antioxidants are generally less potent than synthetic alternatives and may require higher inclusion or shorter shelf-life assumptions.⁵
Labeling and Disclosure Limitations
Preservative labeling is governed by ingredient disclosure rules rather than functional transparency.
Important considerations include:
Preservatives added by ingredient suppliers may not appear on final labels
“Preserved with mixed tocopherols” does not specify oxidation stage or effectiveness
Label claims do not reflect oxidative load at time of feeding
As a result, labels describe intended preservation, not real-time food condition.
Preservation vs. Nutrient Integrity
While preservatives slow fat oxidation, they do not prevent nutrient degradation caused by heat processing.
Preservation:
Maintains shelf stability
Supports palatability over time
Protects fats from rapid rancidity
Preservation does not:
Restore heat-damaged proteins
Replace destroyed enzymes
Reverse vitamin loss during extrusion
This distinction explains why preservation and fortification are separate requirements in kibble production.⁶
Shelf Life and Industrial Distribution
Dry pet food supply chains often require:
12–24 months of shelf stability
Transport across variable climates
Storage under non-refrigerated conditions
Preservatives allow kibble to function as a globally distributable industrial product rather than a perishable food.⁷
Regulatory Oversight of Preservatives
Regulatory agencies evaluate preservatives for safety at defined inclusion levels, not for long-term dietary optimization.
AAFCO and FDA frameworks:
Approve preservatives based on toxicological thresholds
Do not assess cumulative dietary exposure across multiple foods
Do not evaluate interactions with processing-induced compounds
Preservatives are assessed as additives, not as components of a biologically appropriate diet.
Stability Over Freshness
Preservatives allow dry pet food to exist as a shelf-stable product.
They slow oxidation, maintain appearance and aroma, and support long distribution cycles. They do not preserve food integrity in the biological sense.
Understanding preservatives clarifies why kibble prioritizes stability and consistency rather than freshness or structural nutrient preservation.
Citations, Sources & Footnotes
Shahidi, F. (2010). “Lipid oxidation and improving the oxidative stability.” Chemical Society Reviews. Accessed February 3, 2026. https://pubs.rsc.org/en/content/articlehtml/2010/cs/b922183m (RSC Publishing)
Aldrich, G. (2018). “How to formulate, launch new pet food and treats” (includes fat application + palatant adherence factors). Petfood Industry. Accessed February 3, 2026. https://www.petfoodindustry.com/pet-food-market/article/15464735/how-to-formulate-launch-new-pet-food-and-treats (PetfoodIndustry)
Frankel, E.N. (2005). Lipid Oxidation (2nd ed.). Elsevier/Woodhead. Accessed February 3, 2026. https://shop.elsevier.com/books/lipid-oxidation/frankel/978-0-9531949-8-8 (Elsevier Shop)
U.S. Food and Drug Administration (FDA). (2025, Feb 13). “Substances Added to Food (formerly EAFUS)” (Food Additive Status List). Accessed February 3, 2026. https://www.fda.gov/food/food-additives-petitions/food-additive-status-list (U.S. Food and Drug Administration)
Pokorný, J. (2001). “Natural antioxidant functionality during food processing.” Trends in Food Science & Technology. Accessed February 3, 2026. https://www.researchgate.net/publication/288930790_Natural_antioxidant_functionality_during_food_processing (ResearchGate)
National Research Council. (2006). Nutrient Requirements of Dogs and Cats. National Academies Press. Accessed February 3, 2026. https://nap.nationalacademies.org/catalog/10668/nutrient-requirements-of-dogs-and-cats (U.S. Food and Drug Administration)
Phillips-Donaldson, D. (2025, Dec 19). “Pet food innovation going in wrong direction for this economy?” Petfood Industry. Accessed February 3, 2026. https://www.petfoodindustry.com/blogs-columns/adventures-in-pet-food/blog/15774569/pet-food-innovation-going-in-wrong-direction-for-this-economy (PetfoodIndustry)

